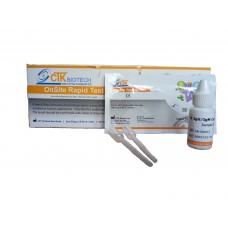
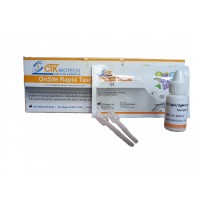
Швидкий тест тропонін (кров, сироватка або плазма) – це імунохроматографічний аналіз для якісного визначення серцевого тропоніну І та його комплексу в сироватці, плазмі або цільній крові людини в концентрації, що дорівнює або вища, ніж 1 нг/мл. Він призначений для використання в якості скринінгового тесту професіоналами, так як допоміжний засіб при діагностиці гострого інфаркту міокарда (ІМ).
Комбінований експрес-тест «ОнСайт» на виявлення тропоніну І може дати тільки попередній результат аналізу, щоб допомогти в діагностиці ГІМ. Будь-яке використання або інтерпретація цього попереднього результату тесту також повинно залежати від симптомів пацієнта і професійної оцінки лікаря. Більш точний тестовий метод, такий як інструментальний метод, повинен бути використаний для підтвердження результату тесту, отриманого за допомогою даного пристрою.
Серцевий тропонін I (cTnI) – це білок, який знаходиться в м’язах серця та має молекулярну масу 22,5 кДа. Разом з тропоніном Т (ТnТ) і тропоніном С (TnC), тропонін І (TnI) утворює комплекс тропоніну в серці, який відіграє фундаментальну роль в передачі внутрішньоклітинних сигналів кальцію, запускаючи взаємодію актоміозину1. cTnI людини має додаткові амінокислотні залишки на його N-кінці, які не існють в формах скелету, таким чином, роблячи cTnI специфічним серцевим маркером 1-3.
Як правило, рівень cTnI в крові дуже низький. cTnI вивільняється в кровоток в формах вільних cTnI і cTnI-С-Т-комплексу через 4-6 годин після пошкодження міокардіальних клітин 2,4,5. Підвищений рівень cTnI може досягати 50 нг/мл протягом 60-80 годин після ГІМ і може бути виявлений протягом 10-14 днів після ГІМ 2,4,5. Таким чином, циркулюючий тропонін І є специфічним і чутливим маркером при діагностиці ГІМ.
Комбінований експрес-тест «ОнСайт» на виявлення тропоніну І призначений для виявлення підвищеного тропоніну I і його комплексу в сироватці, плазмі або цільній крові людини менш ніж за 15 хвилин мінімально кваліфікованим персоналом без необхідності лабораторного обладнання.
Комбінований експрес-тест «ОнСайт» на виявлення тропоніну І – це хроматографічний імуноаналіз. Тестова касета складається з: 1) прокладки для кон’югата бардового кольору, що містить моноклональні антитіла проти тропоніна І, які поєднані з колоїдним золотом (кон'югати антитіл), 2) нітроцелюлозної мембранної смуги, яка містить тестову лінію (смужка T) і контрольну лінію (смужка С). Смужка Т попередньо покривається іншими антитілами проти тропоніна І, а смужка C попередньо покривається антитілом проти контрольної лінії.
Коли достатній обсяг досліджуваного зразка поміщають в лунку для проб тестової касети, зразок мігрує під дією капілярних сил через касету. Підвищений рівень тропоніну I, якщо він присутній в зразку, буде взаємодіяти з кон’югатами антитіл. Потім імунокомплекс фіксується на мембрані, яка попередньо вкрита антитілами проти тропоніна І, утворюючи смужку Т бордового кольору, яка вказує на позитивний результат тесту на виявлення тропоніну І.
Відсутність смужки Т вказує на негативний результат. Тест містить внутрішній контроль (смужку C), яка повинна проявляти бордову лінію імунокомплексу кон’югатів антитіл до контрольної лінії, незалежно від наявності в зразку тропоніну І. Якщо смужка C не розвивається, результат тесту є недійсним, і зразок повинен бути повторно протестований іншим пристроєм.
Рекомендовані товари
Швидкий тест сифіліс (сироватка, плазма)
Швидкий тест на сифіліс є надійним імуноферментним абсорбентом на твердій основі для якісного в..
120.00 грн
Швидкий тест Хелікобактер пілорі (фекалії)
OnSite Експрес-тест на виявлення антигенів бактерії Хелікобактер пілорі – це імунохроматографічний а..
200.00 грн
Швидкий тест Хелікобактер пілорі (сыроватка, плазма, кров)
OnSite Експрес-тест на виявлення антигенів бактерії Хелікобактер пілорі – це імунохроматографічний а..
200.00 грн
Швидкий тест тропонін
Швидкий тест тропонін (кров, сироватка або плазма) – це імунохроматографічний аналіз для якісного ви..
200.00 грн
Швидкий тест холера (фекалії)
Швидкий тест на холеру (фекалії) є надійним імуноферментним абсорбентом на твердій основі для я..
400.00 грн
Швидкий тест гонорея (мазок)
Швидкий тест на гонорею (мазок) є надійним імуноферментним абсорбентом на твердій основі для як..
180.00 грн
Швидкий тест ротавірус (фекалії)
Швидкий тест на ротавірус (фекалії) є надійним імуноферментним абсорбентом на твердій..
180.00 грн
Швидкий тест рота аденовірус (фекалії)
Швидкий тест на рота аденовірус (фекалії) є надійним імуноферментним абсорбентом на т..
180.00 грн
Швидкий тест гепатит В (кров, сироватка, плазма)
Швидкий тест гепатит В (кров, сироватка, плазма) HBsAg-тест-МБАТест-набір імунохроматографічний..
200.00 грн
Швидкий тест гепатит В (сироватка, плазма)
Швидкий тест гепатит В (сироватка, плазма) Комбі-HBV-тест-МБАТест-набір імунохроматографічний д..
200.00 грн
Швидкий тест гепатит С (кров, сироватка, плазма)
Швидкий тест гепатит С (кров, сироватка, плазма) АТ-ВГС-тест-МБАТест-набір імунохроматографічни..
150.00 грн
Швидкий тест гепатит В (сироватка, плазма)
Швидкий тест гепатит В (сироватка, плазма) HBеAg-тест-МБАТест-набір імунохроматографічний для в..
200.00 грн
Швидкий тест гепатит В (сироватка, плазма)
Швидкий тест гепатит В (сироватка, плазма) HBеAg-тест-МБАТест-набір імунохроматографічний для в..
200.00 грн
Швидкий тест гепатит А (сироватка, плазма)
Швидкий тест гепатит А (сироватка, плазма) АТ-ВГА-тест-МБАТест-набір імунохроматографічний для ..
180.00 грн
Комбінований тест на TORCH IgM/IgG (токсоплазмоз, краснуха, цитомегаловірус, ВПГ-1, ВПГ-2) (у цільній крові/сироватці/плазмі)
Комбінований тест на TORCH IgM/IgG (токсоплазмоз, краснуха, цитомегаловірус, ВПГ-1, ВПГ-2)..
300.00 грн
Швидкий тест ВІЛ, гепатит В, гепатит С (сироватка, плазма)
Швидкий тест ВІЛ, гепатит В, гепатит С (сироватка, плазма) Мульти 3-тест-МБАТес..
180.00 грн
Швидкий тест гепатит В (сироватка, плазма)
Швидкий тест мікоплазма (сироватка, плазма) Міко-тест-МБАТест-набір імунохроматографічний для в..
200.00 грн
Швидкий тест гонорея (мазок, секрет)
Швидкий тест гонорея (мазок, секрет) Гоно-тест-МБАТест-набір імунохроматографічний для виявленн..
170.00 грн
Швидкий тест хламідія (зішкріб)
Швидкий тест хламідія (зішкріб) Хламі-тест-МБАТест-набір імунохроматографічний для виявлення хл..
170.00 грн
Швидкий тест сальмонела (кров, сироватка, плазма)
Швидкий тест сальмонела (кров, сироватка, плазма) Сальмонела-IgG/IgM-тест-МБАТест-набір імунохр..
200.00 грн
Швидкий тест туберкульоз (сироватка, плазма, кров)
Швидкий тест на туберкульоз (сироватка, плазма, кров) є надійним імуноферментним абсорбен..
200.00 грн
Швидкий тест стрептокок групи А (мазок)
Швидкий тест стрептокок групи А (мазок) Strep A-тест-МБАТест-набір імунохроматографіч..
170.00 грн
Швидкий тест аденовірус (фекалії)
Швидкий тест аденовірус (фекалії) Адено-тест-МБАТест-набір імунохроматографічний для виявлення&..
150.00 грн
Швидкий тест малярія (кров)
Швидкий тест малярія (кров) Малярія-тест-МБАТест-набір імунохроматографічний для виявлення..
190.00 грн
Швидкий тест ТORCH (сироватка, плазма)
Швидкий тест цитомегаловірус, токсоплазмоз, вірус краснухи, герпес 1 та 2 типів (сироватка..
300.00 грн
Швидкий тест токсоплазма (сироватка, плазма)
Швидкий тест токсоплазма (сироватка, плазма) Токсо-тест-МБАТест-набір імунохроматографічн..
180.00 грн
Швидкий тест цитомегаловірус (сироватка, плазма)
Швидкий тест цитомегаловірус (сироватка, плазма) ЦМВ-тест-МБАТест-набір імунохроматог..
200.00 грн
Швидкий тест краснуха (сироватка, плазма)
Швидкий тест краснуха (сироватка, плазма) Краснуха-тест-МБАТест-набір імунохроматографічни..
170.00 грн
Швидкий тест герпес 1 типу (сироватка, плазма)
Швидкий тест герпес 1 типу (сироватка, плазма) ВПГ1-тест-МБАТест-набір імунохроматографічн..
150.00 грн
Швидкий тест герпес 2 типу (сироватка, плазма)
Швидкий тест герпес 2 типу (сироватка, плазма) ВПГ2-тест-МБАТест-набір імунохроматографічн..
150.00 грн